Old Blood Stem Cells Regain Youthful Function in Mice
Introduction
A recent Cell Stem Cell study reports that aged blood-forming stem cells can recover several youthful features when scientists correct dysfunction in lysosomes, the tiny recycling structures inside cells [1]. The finding was highlighted in a ScienceDaily report from Mount Sinai, which described the work as making old blood stem cells “act young again” in mice [2]. The study’s central claim is more precise: aged hematopoietic stem cells, or HSCs, develop lysosomes that become hyperacidic, damaged, depleted, and abnormally active; suppressing this overactivity restored lysosomal integrity, improved metabolism and epigenetic stability, reduced inflammatory signaling, and increased the cells’ ability to repopulate blood in living animals [1].
Hematopoietic stem cells are rare, long-lived cells in the bone marrow that generate the body’s blood and immune cells [2]. When these cells age, they typically lose regenerative strength, become biased toward producing certain blood cell types, and show more inflammatory behavior [1]. This matters because the blood system is not just about red blood cells. It also supports immune defense, tissue repair, and protection from infection. A weaker blood-forming system can contribute to poorer immune responses, higher inflammatory burden, and age-related vulnerability [2].
This study does not show that human aging has been reversed. It does not prove that people can take a lysosome-targeting drug to become biologically younger. Instead, it shows that in aged mouse blood stem cells, lysosomal dysfunction appears to be a major and targetable driver of stem cell aging [1]. The most striking quantitative result was that ex vivo treatment of old HSCs with a lysosomal inhibitor boosted their in vivo blood-forming, or repopulation, capacity by more than eightfold [1].
In simpler terms, the researchers removed old blood stem cells, treated them in the lab to calm overactive lysosomes, and then tested whether the cells worked better in living animals. The treated cells behaved more like young stem cells in several important ways.
What the Study Asked
The paper asked whether lysosomes are passive cleanup compartments in aging blood stem cells or active drivers of age-related decline [1]. Lysosomes are often described as cellular recycling centers because they break down proteins, nucleic acids, carbohydrates, lipids, and damaged cellular material so the cell can reuse the parts [2]. They also help regulate nutrient availability and metabolism, meaning they influence how cells build, break down, and respond to stress [2].
The researchers focused on a specific problem in aged HSCs: older blood stem cells often lose the ability to renew themselves and generate balanced blood and immune cell populations [1]. Previous research has connected HSC aging with inflammation, mitochondrial stress, clonal hematopoiesis, and higher risk of myeloid malignancies, but the role of lysosomes has been less clear [1]. This study tested whether lysosomal dysfunction sits upstream of these aging features.
The key research question was: if lysosomal overactivity is corrected, can old blood stem cells regain youthful function? The answer, based on the reported mouse and cellular data, was yes for several stem-cell features, including lysosomal integrity, metabolic homeostasis, inflammatory signaling, self-renewal, and repopulation capacity [1].
Why This Research Matters
Blood-forming stem cells are foundational to healthy aging because they continually replenish blood and immune cells throughout life [2]. When HSCs age, the immune system can become less resilient. Older adults often face weaker responses to infections and more inflammation, and HSC aging is also linked with clonal hematopoiesis, an age-associated condition in which certain blood-cell clones expand and can raise risk for blood cancers and inflammatory disease [2].
The Mount Sinai report explains that aging HSCs lose their capacity to renew and repair the blood system, which can reduce the body’s ability to fight infections [2]. The article also notes that clonal hematopoiesis becomes more common with age and is considered a premalignant state associated with blood cancers and inflammatory disorders [2]. The Cell Stem Cell abstract likewise links HSC aging with clonal hematopoiesis, myeloid malignancies, and immune decline [1].
The study matters because it identifies a potentially reversible mechanism. Many age-related changes are treated as gradual wear-and-tear processes. Here, the authors argue that old HSCs are not simply exhausted beyond repair. Their lysosomes are malfunctioning in a way that can be targeted experimentally [1]. When lysosomal overactivity was suppressed, old HSCs recovered features that looked more youthful, including improved metabolism, lower inflammatory signaling, better epigenetic balance, and stronger regenerative function [1].
This is an important distinction. The work does not claim that every feature of organism-wide aging can be reversed. It suggests that at least one old stem-cell population retains recoverable capacity when a specific cellular stress pathway is corrected.
How the Study Was Done
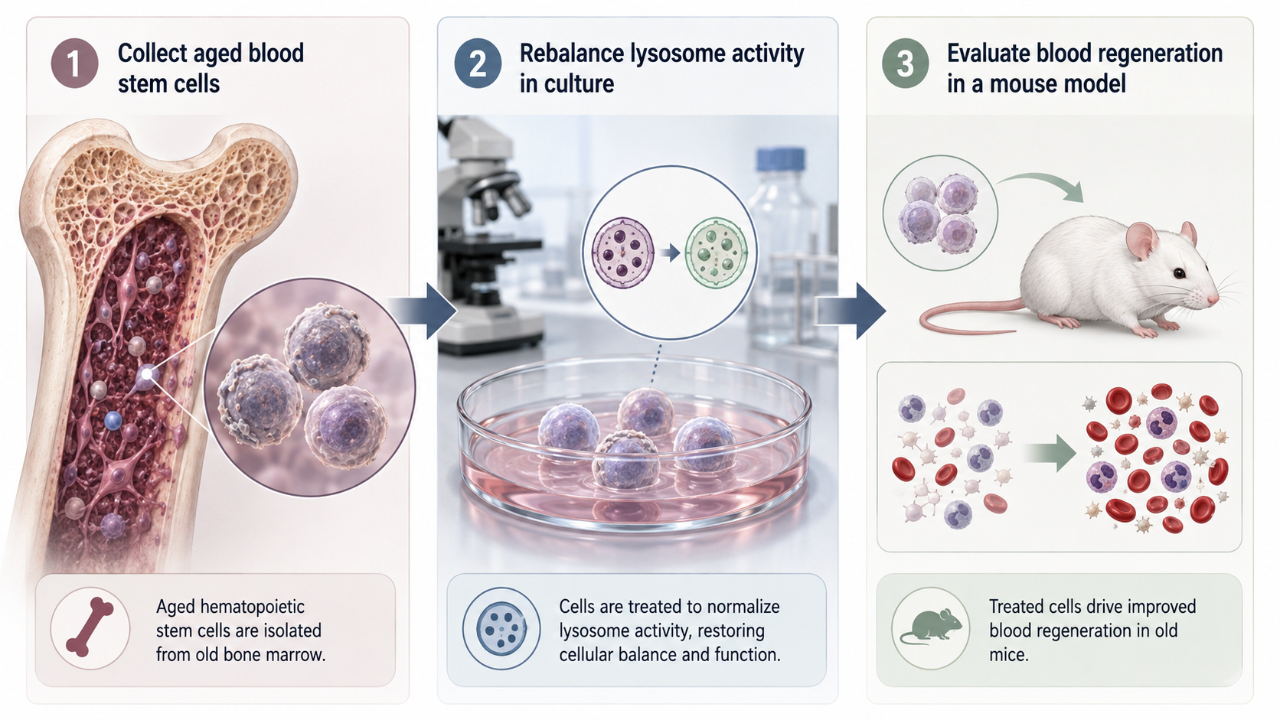
The full paper text was not provided in the input, so some details—such as exact animal numbers, dosing conditions, statistical methods, and all control groups—cannot be fully assessed here. The available abstract, institutional summary, and ScienceDaily report describe several major methods [1, 2].
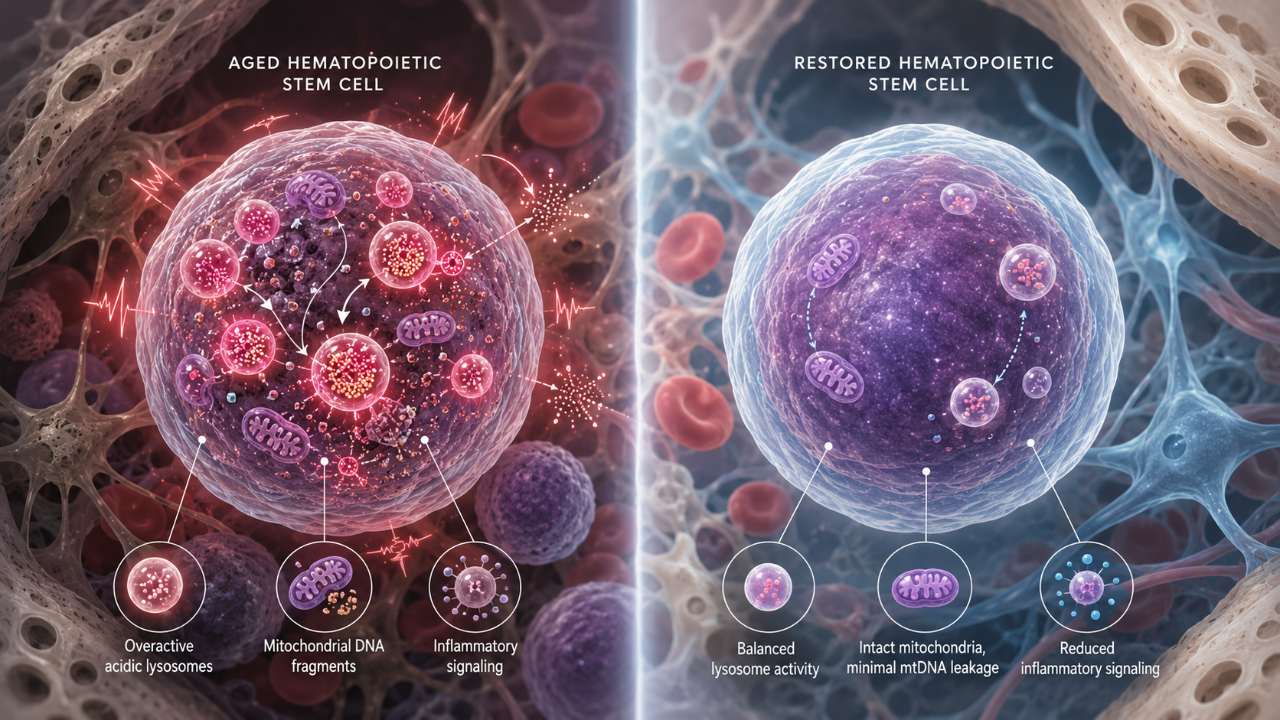
First, the team studied aged hematopoietic stem cells and compared their cellular state with younger or healthier stem-cell behavior [1]. They found that lysosomes in aged HSCs were hyperacidic, depleted, damaged, and aberrantly activated [1]. In practical terms, the recycling system inside these old stem cells appeared to be running in a stressed and dysfunctional mode rather than a balanced maintenance mode.
Second, the researchers used single-cell transcriptomics, a method that reads gene-expression patterns cell by cell [1]. This helps reveal whether a stem-cell population is uniform or whether different cells are following different inflammatory, metabolic, or aging-related programs. Single-cell methods are useful in stem-cell aging because old stem-cell pools can become heterogeneous: some cells may be more damaged, more inflammatory, or more functionally impaired than others.
Third, the researchers used functional assays to test whether the cells could actually regenerate blood, not merely look better in molecular measurements [1]. This distinction matters. A cell can show a healthier gene-expression pattern without necessarily performing well in a living organism. The study reports that ex vivo lysosomal inhibition improved in vivo repopulation capacity by more than eightfold, meaning treated old HSCs generated blood more effectively after transplantation into living animals [1].
Fourth, the team used a vacuolar ATPase, or v-ATPase, inhibitor to suppress lysosomal hyperactivation [1]. v-ATPase helps acidify lysosomes. Because aged HSC lysosomes were described as hyperacidic and overactive, dampening this pathway helped restore lysosomal function and downstream cellular balance [1].
The overall experimental logic was straightforward: identify an aging-associated defect, intervene on that defect, and then test whether old stem cells recover meaningful function. The strongest part of the study is that it links a mechanism—lysosomal dysfunction—to functional regeneration rather than stopping at molecular description.
Key Findings
The study’s first major finding was that aged HSCs have lysosomes that are not merely old or inefficient, but abnormally activated. The paper describes aged HSC lysosomes as hyperacidic, depleted, damaged, and aberrantly activated [1]. This suggests that the aging defect is not a simple lack of cellular cleanup. Instead, the cleanup system itself appears to become overdriven and destabilizing.
The second major finding was that suppressing lysosomal hyperactivation restored lysosomal integrity and improved metabolic and epigenetic homeostasis in old HSCs [1]. “Metabolic homeostasis” refers to a cell’s ability to manage energy, nutrients, and molecular building blocks in a stable way. “Epigenetic homeostasis” refers to the organization of chemical signals that help regulate which genes are turned on or off without changing the DNA sequence.
The third major finding was that correcting lysosomal dysfunction reduced inflammatory and interferon-driven programs [1]. Interferon-related signaling is part of immune defense, especially against viruses, but chronic activation can contribute to inflammation and cellular stress. The paper connects this inflammatory reduction to better lysosomal processing of mitochondrial DNA and reduced activity of the cGAS-STING pathway [1]. The cGAS-STING pathway is a cellular alarm system that detects misplaced DNA and can trigger inflammatory immune responses.
A helpful way to think about this mechanism is that damaged or poorly processed mitochondrial DNA can look like a danger signal inside the cell. If lysosomes cannot handle that material properly, the cell may activate immune alarms even when there is no infection.
The fourth major finding was functional. Ex vivo lysosomal inhibition increased old HSCs’ in vivo repopulation capacity by more than eightfold and improved self-renewal [1]. This is the result that moves the study from “cells looked younger” to “cells performed better.” In blood stem-cell biology, repopulation capacity is a central measure of whether HSCs can rebuild blood lineages after transplantation.
The fifth major finding was broader rejuvenation-like change. The ScienceDaily and Mount Sinai summaries report that treated old stem cells produced more balanced blood and immune cells, improved mitochondrial performance, showed healthier epigenetic patterns, reduced inflammation, and generated fewer damaging inflammatory signals [2, 3].
Key Findings Table
| Finding | What changed in aged HSCs | Why it matters | Evidence strength |
|---|---|---|---|
| Lysosomes were abnormal | Hyperacidic, depleted, damaged, and overactivated | Identifies a concrete cellular defect associated with HSC aging | High for the studied model [1] |
| Lysosomal inhibition restored function | v-ATPase inhibition improved lysosomal integrity | Shows the defect was targetable, not merely descriptive | High for preclinical HSC assays [1] |
| Inflammation decreased | Interferon-driven programs and cGAS-STING signaling were reduced | Links lysosomes, mitochondrial DNA processing, and inflammatory aging | Moderate to high within the study model [1] |
| Regeneration improved | Old HSC repopulation capacity increased more than eightfold | Demonstrates functional recovery in living animals | High for mouse/preclinical transplantation assays [1] |
| Human therapy remains unproven | No clinical trial in people is reported in the provided source | Prevents overextension of the finding | High caution [1, 2] |
Suggested visual: a simple mechanism diagram showing aged HSCs with overactive lysosomes, poor mitochondrial DNA processing, cGAS-STING inflammation, and weak blood regeneration; then treated HSCs with calmer lysosomes, lower inflammatory signaling, and stronger blood regeneration.
What the Results Mean
The study suggests that lysosomal dysfunction is not just a byproduct of HSC aging; it may be one of the drivers [1]. This is important because driver mechanisms are more useful therapeutic targets than passive markers. If a feature merely reflects aging, changing it may not help. If it contributes to aging, correcting it can restore function.
The reported effect is especially notable because the intervention was performed ex vivo [1]. That means old stem cells were treated outside the body before being tested in living animals. Ex vivo approaches can sometimes be more controllable than systemic treatments because the target cells can be modified or conditioned in a laboratory setting. However, ex vivo treatment also raises practical hurdles: collecting stem cells, treating them safely, validating the result, and returning them without causing harm are complex steps.
The study also connects several important aging pathways into one chain. In aged HSCs, lysosomal overactivity appears to impair processing of mitochondrial DNA [1]. Poorly processed mitochondrial DNA can activate cGAS-STING signaling, which can trigger inflammatory programs [1]. Those inflammatory programs can weaken stem-cell health and regenerative function. By restoring lysosomal balance, the researchers reduced that inflammatory cascade and improved stem-cell performance [1].
This does not mean lysosomes are the only cause of blood stem-cell aging. Aging is multi-layered. DNA damage, epigenetic drift, metabolic stress, inflammation, immune remodeling, and clonal expansion can all contribute. The stronger interpretation is that lysosomal dysfunction may be a key upstream node in aged HSCs—one that connects cleanup failure, mitochondrial stress, inflammation, and regenerative decline.
Strengths of the Study
A major strength is that the study combines molecular analysis with functional testing. Single-cell transcriptomics can reveal how old stem cells differ from young ones, but gene-expression data alone do not prove that a cell can regenerate tissue [1]. The reported transplantation-style functional assays add weight because they test whether treated old HSCs can actually repopulate blood in living animals [1].
Another strength is the mechanistic depth. The study does not simply report that lysosomes look abnormal. It connects lysosomal hyperactivation to mitochondrial DNA handling, cGAS-STING signaling, inflammatory programs, metabolic homeostasis, epigenetic state, and self-renewal [1]. That chain makes the finding more biologically persuasive.
The magnitude of the functional effect is also substantial. A more than eightfold boost in in vivo repopulation capacity after ex vivo lysosomal inhibition is a large signal in the context of aged HSC function [1]. Large effects still need replication, but they provide a strong reason for further investigation.
The study also appears to focus on a highly relevant cell type. HSCs sit at the root of blood and immune-cell production [2]. A therapy that improves their function could eventually matter for immune aging, transplant biology, inflammatory disease, and possibly age-related blood disorders. Those applications remain future possibilities rather than proven outcomes.
Limitations and Cautions
The most important limitation is that this is preclinical research. The provided material describes aged HSCs, functional assays, and living-animal repopulation testing, but it does not describe a human clinical trial [1, 2]. The result should therefore be interpreted as a promising biological discovery, not a ready anti-aging therapy.
A second limitation is incomplete access to the full paper in the provided input. The available material includes the paper metadata, abstract-level details, and institutional summaries, but not the complete methods, all figures, all statistical tests, or supplementary methods. That means some details cannot be assessed here, including exact animal numbers, randomization procedures, blinding, treatment dose, duration, sex distribution, and all controls.
A third caution concerns the intervention itself. v-ATPase inhibition can affect lysosomal acidification, but lysosomes are essential in many cells [1]. Broadly suppressing lysosomal function in a whole organism could create unintended effects. The reported approach involved ex vivo treatment of old HSCs, which may reduce some systemic risks, but translation to human cells would require careful safety testing.
A fourth limitation is that rejuvenation is being used in a specific cellular sense. The study reports restored youthful state in aged HSCs, including improved metabolic, epigenetic, inflammatory, and regenerative features [1]. That is not the same as reversing organism-wide aging. A person’s aging biology involves many tissues, immune history, vascular function, brain function, metabolism, and environmental exposures.
What This Does Not Prove
This study does not prove that aging can be reversed in humans. It does not prove that lysosomal inhibitors can safely rejuvenate people. It does not show that taking a drug can prevent blood cancers, restore immune function in older adults, or extend lifespan. The paper supports a narrower and more defensible conclusion: in aged mouse blood stem cells, lysosomal hyperactivation and dysfunction appear to drive important features of HSC aging, and correcting that dysfunction can restore several youthful functions in experimental systems [1].
The study also does not prove that all aged stem cells behave the same way. HSCs have specialized biology. Other stem-cell types—such as neural, muscle, skin, or intestinal stem cells—may have different aging mechanisms. Lysosomes may still matter in those cells, but this paper’s direct evidence concerns aged HSCs [1].
Finally, the study does not eliminate the complexity of clonal hematopoiesis or myeloid malignancy risk. It links HSC aging to conditions such as clonal hematopoiesis, immune decline, and myeloid malignancies, and it identifies a mechanism that may contribute to the aging state [1, 2]. It does not demonstrate prevention or treatment of those conditions in humans.
Broader Implications
The research supports a growing view that some aging features are plastic rather than fixed. In this context, plastic means old cells can sometimes recover function if the right stress pathway is corrected. The study’s language—“restores youthful state”—reflects multiple levels of recovery: lysosomal integrity, metabolism, epigenetic balance, lower inflammatory signaling, self-renewal, and blood-forming capacity [1].
For regenerative medicine, the ex vivo aspect is especially interesting. In principle, patient-derived cells could be treated outside the body and then returned, similar in concept to some cell therapies. That idea remains speculative for this specific approach, but the more-than-eightfold improvement in old HSC repopulation capacity provides a rationale for deeper preclinical work [1].
For aging biology, the study elevates lysosomes from background cleanup organelles to active regulators of stem-cell aging. It suggests that aging research should pay close attention not only to DNA damage and inflammation, but also to how cells process waste, handle mitochondrial DNA, and tune immune alarm pathways.
For public understanding, the main takeaway should be balanced. This is a meaningful step in cellular rejuvenation research. It is not an anti-aging treatment for people. It shows that old blood stem cells in mice can recover youthful behavior under controlled experimental conditions when a specific lysosomal defect is corrected [1, 2].
Terms Worth Knowing
| Term | Plain-language meaning |
|---|---|
| Hematopoietic stem cells (HSCs) | Bone marrow stem cells that produce blood and immune cells |
| Lysosome | A cellular recycling structure that breaks down and reuses biological material |
| v-ATPase | A protein complex that helps acidify lysosomes |
| Ex vivo | A process done outside the body, often on cells removed from an organism |
| In vivo | A process tested inside a living organism |
| cGAS-STING pathway | A cellular alarm system that detects misplaced DNA and triggers inflammatory signaling |
| Epigenetic homeostasis | Stable regulation of gene activity without changing DNA sequence |
| Repopulation capacity | The ability of transplanted stem cells to rebuild blood-cell populations |
Balanced Critical Appraisal
The evidence is strong that lysosomal dysfunction is a major feature of aged HSCs in the reported model and that targeted lysosomal suppression can improve old HSC function in preclinical assays [1]. The use of single-cell transcriptomics plus functional repopulation testing makes the study more persuasive than a purely descriptive aging paper [1].
The evidence is moderate for broader claims about anti-aging therapy. The ScienceDaily headline emphasizes “old blood stem cells young again,” which captures the central result but can sound broader than the data support [2]. The more careful interpretation is that aged HSCs recovered youthful characteristics in experimental systems after lysosomal dysfunction was corrected [1]. That is still important. It is also narrower than a therapy for human aging.
The evidence is low or absent for clinical application at this stage. The provided sources do not describe human testing, long-term safety, clinical dosing, or therapeutic outcomes in older adults. The next scientific step is not consumer use; it is replication, mechanistic refinement, testing in human HSCs, safety profiling, and determining whether the approach can improve clinically meaningful outcomes without disrupting essential lysosomal biology.
Final Summary
The Cell Stem Cell study identifies lysosomal dysfunction as a key driver of aging in hematopoietic stem cells, the bone marrow cells that generate blood and immune cells [1]. In aged HSCs, lysosomes became hyperacidic, damaged, depleted, and overactive [1]. This dysfunction appears to disturb metabolism and epigenetic stability while feeding inflammatory pathways through impaired mitochondrial DNA processing and cGAS-STING activation [1].
When the researchers suppressed lysosomal hyperactivation using a v-ATPase inhibitor, aged HSCs recovered several youthful features: healthier lysosomes, improved metabolism, reduced inflammatory signaling, better self-renewal, and stronger blood-forming capacity [1]. The standout result was a more than eightfold improvement in in vivo repopulation capacity after ex vivo treatment of old stem cells [1].
The report is legitimately exciting for stem-cell aging research, but it should be read carefully. It does not show human age reversal. It does not establish a treatment for aging, immune decline, or blood cancers. It shows that old blood-forming stem cells retain a capacity to recover when a specific lysosomal stress pathway is corrected. That makes lysosomes a serious target for future regenerative-medicine research and a clearer lens for understanding how blood aging may be driven from inside the stem cell itself [1].
References
- Arif, T., Qiu, J., Khademian, H., Lohithakshan, A., Menon, A., Menon, V., Slavinsky, M., Batignes, M., Lin, M., Sebra, R., Beaumont, K. G., Benson, D. L., Tzavaras, N., Ménager, M. M., & Ghaffari, S. (2025). Reversing lysosomal dysfunction restores youthful state in aged hematopoietic stem cells. Cell Stem Cell, 32(12), 1904–1922.e7. https://doi.org/10.1016/j.stem.2025.10.012
- The Mount Sinai Hospital / Mount Sinai School of Medicine. (2026, May 12). Scientists make old blood stem cells young again in major anti-aging breakthrough. ScienceDaily. https://www.sciencedaily.com/releases/2026/05/260511213204.htm
- Icahn School of Medicine at Mount Sinai. (2026). Restoring a youthful state to aging blood cells. https://icahn.mssm.edu/research/research-saves-lives/restoring-aging-blood-cells